
Live-cel Technology
The programs at Glafabra Therapeutics target a variety of enzyme deficiency disorders with our novel, long-lasting Live-cel therapy. We are bringing cell-based gene therapies to patients with Fabry, Pompe and Gaucher disease. Clinically demonstrated in Fabry to last at least 5 years from one dose, our technology provides durable solutions to increase the freedom in the lives of those suffering from debilitating enzyme disorders.
Clinical Stage
Our Live-cel therapy approach is an improvement over intermittent enzyme exposure with enzyme replacement therapy (ERT). Unlike traditional ERT, our Live-cel therapy provides an uninterrupted supply of enzyme. The durability of our Live-cell therapy is now demonstrated to exceed 5 years (PMID 39794302). This indicates the hassle of infusions done every other week can be now be turned into one infusion every 5 years - a 130x increase in patient freedom. Furthermore, the transplanted leukocytes are known to spread systemically to a wide variety of compartments in the body, including the brain, thus the blood brain barrier problem with traditional recombinant protein transfusions is a much lower obstacle. As a result, successful treatment of neurological complications of enzyme deficiency can be expected.
How it works
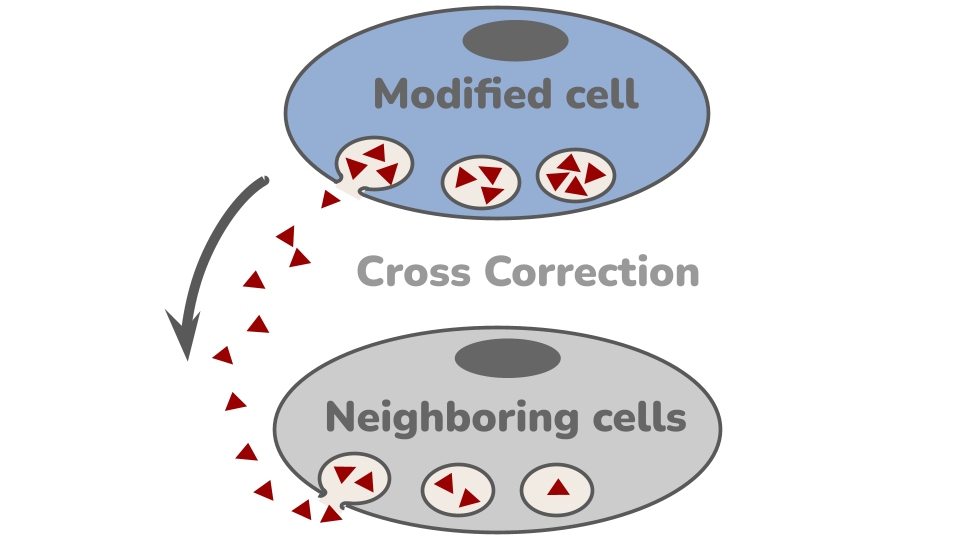
The Live-cell therapy is made by are harvested from stem cells from a patient and then genetically engineering them to make the missing enzyme. These modified cells are then put back into the body as the therapy, where they engraft and make the white blood cell progeny. These white blood cells go everywhere in the body and start to secrete the missing enzyme. In a process called Cross Correction, neighboring cells can take up the enzyme and use it to stop the build up of toxic lipid metabolites.
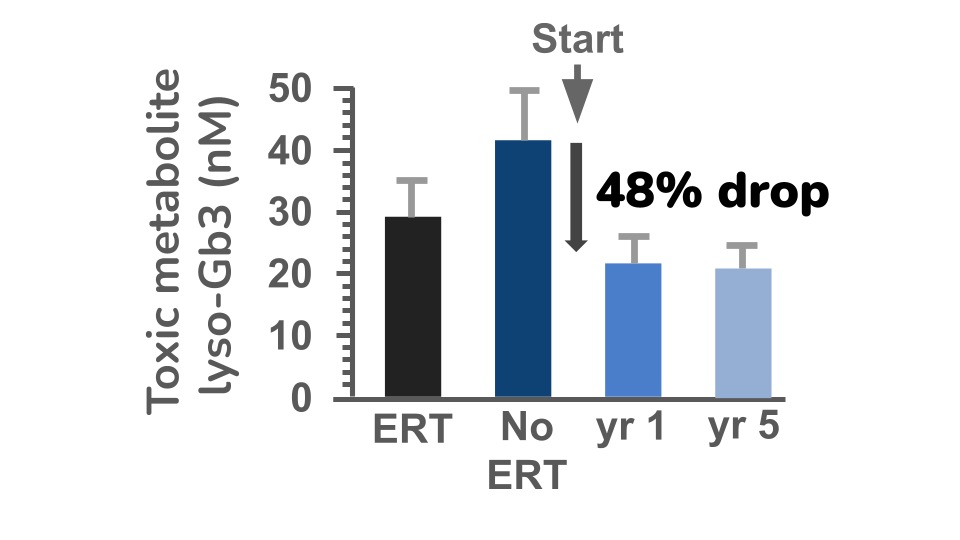
Our lead program is GT-GLA-S03 Live-cel therapy for Fabry disease and it was evaluated in clinical trials in Canada. To measure how well the treatment worked, the Canadian researchers measured the build up of the harmful lipid (fatty) substance in the blood called lyso-Gb3, which acts as a biomarker for disease severity. At the start of the study, all patients were receiving standard enzyme replacement therapy (ERT) and their lyso-Gb3 levels were at a steady and safe plateau level . When ERT was stopped, levels of lyso-Gb3 increased (No-ERT), confirming that the disease activity was rising again. After patients received the cell therapy, lyso-Gb3 levels decreased by 48%, showing a substantial reduction in harmful buildup. Importantly, a reassessment of patients, five years after their initial dose of cell therapy, the lyso-Gb3 levels remained well controlled - no build up occurred. This indicates Live-cel therapy can provide at least a 130x increase in durability treatment from a single dose, when compared to standard of care with ERT. As a result, patients receiving Live-cel will obtain a dramatic improvement in quality of their lifestyle.
“The benefits were many. I have felt the best I have felt in my life”
- patient after receiving GT-GLA-S03
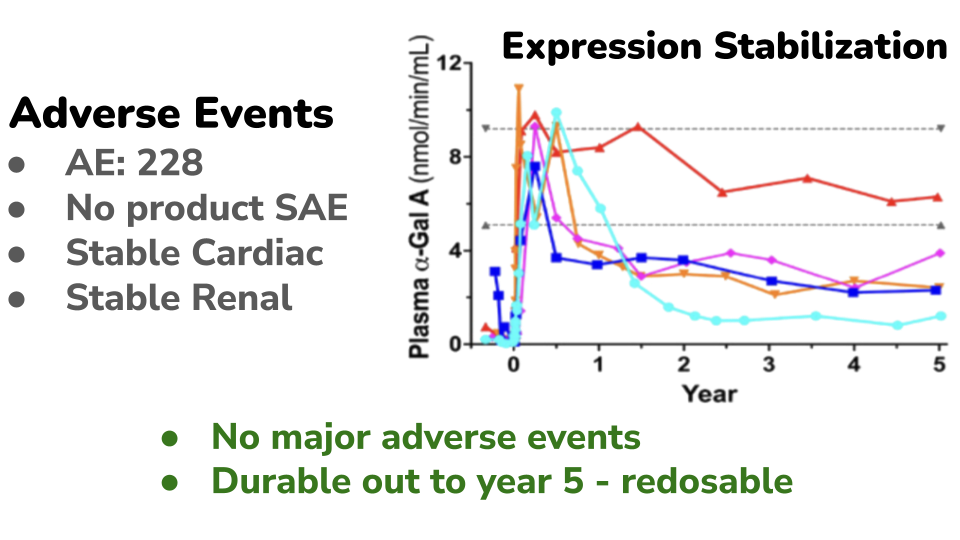
The therapy has demonstrated a favorable safety profile in clinical evaluation (PMID 39794302). Across the study, 228 adverse events (AEs) were reported, the majority of which were mild to moderate. Only 20 AEs were classified as Grade 3–4 (more serious). Importantly, no severe adverse events (SAEs) were determined to be related to the cell therapy product. Key organ function measures, including cardiac and renal parameters, remained stable over five years of follow-up. This is particularly important given these are the organ systems typically affected by the disease. Following treatment under an outpatient conditioning protocol, patients experienced an initial rise in enzyme activity. As expected in cell-based therapies, this early peak gradually declined and stabilized at a sustained level over time. A similar pattern was observed in vector copy number (VCN), a measure of how many therapeutic gene copies are present in treated cells (data not shown). VCN initially peaked at approximately 1 copy per cell and then stabilized at around 0.2 copies per cell by the second year after treatment. This stabilized level remained durable over long-term follow-up. Based on these observations, repeat dosing may provide additional benefit for some patients. A second treatment could potentially increase the stabilized VCN baseline, which may enhance long-term enzyme production and therapeutic effect.
Our platform technology can be applied to a variety of other diseases where therapeutic cross-correction can provide patient benefit. Contact us for more information.
